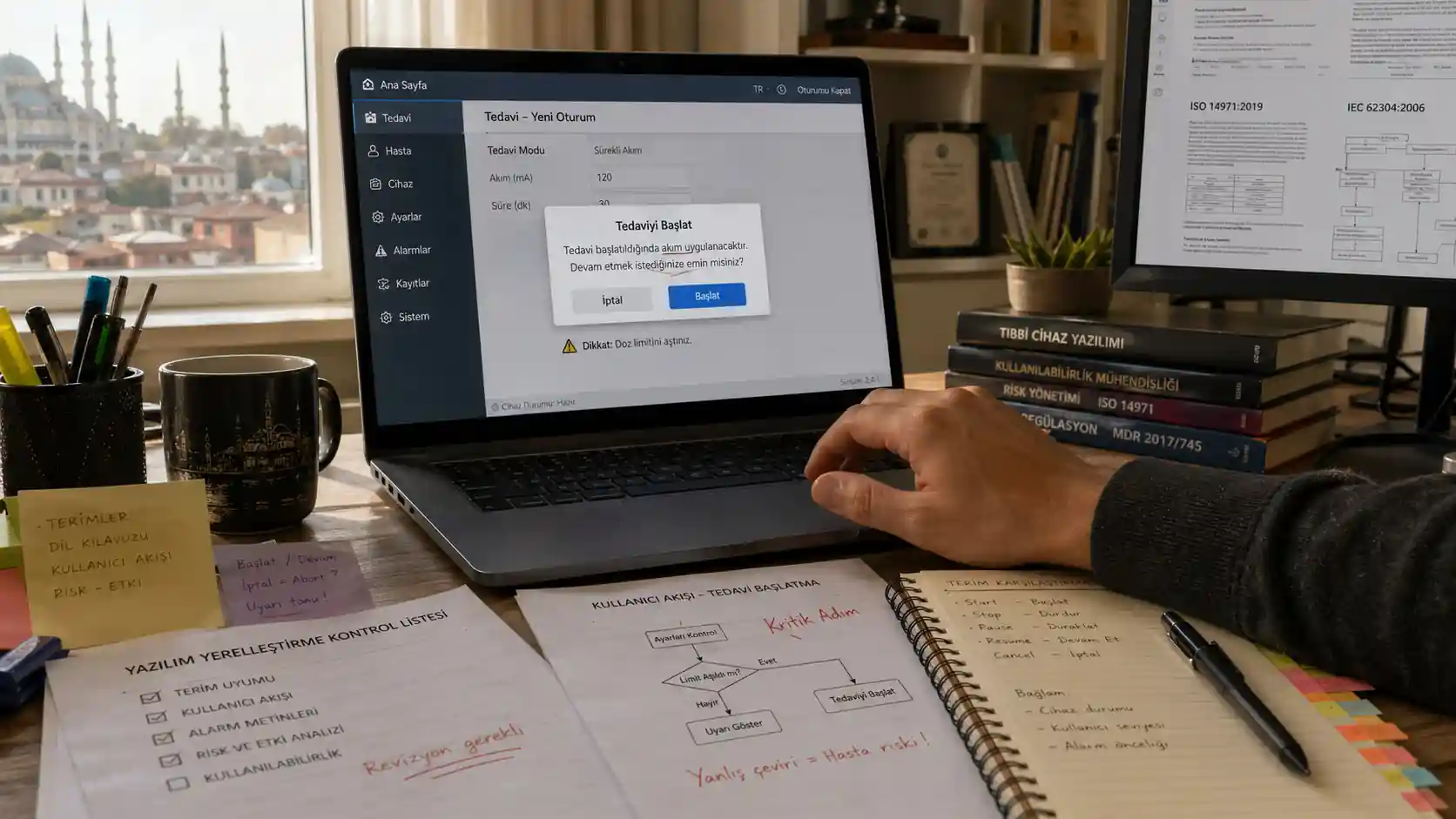
A Turkish UI Label Can Look Small Until It Changes Clinical Behavior.
Medical device software language must preserve safety, sequence, responsibility, and user action in Turkish.
Turkish Medical Device Software Localization: Why UI Wording Becomes a Patient Safety Risk
A medical device software interface reaches Turkish users. The product works. The clinical workflow has been validated. The software has already passed multiple internal reviews.
Then a user reaches a warning screen.
The Turkish label is short. The button fits. The message is grammatical. But it is not quite clear whether the user should stop, confirm, repeat, review, or continue.
That small hesitation can become a safety issue.
In medical software, localization is not a cosmetic layer. It is part of how users act.
What Actually Breaks
Medical device software localization often fails because UI strings are treated separately from clinical workflow. A label is exported from the interface, translated in a spreadsheet, then reinserted into the product.
That workflow hides context. The translator may not see what comes before the message, what happens after the user clicks, or whether the screen appears during a normal procedure, warning state, calibration step, error state, or clinical review.
The result can be a Turkish string that is linguistically acceptable but clinically weak. It tells the user something, but not enough to support safe action.
The most dangerous failures happen in short text: button labels, status messages, warnings, measurement descriptions, and confirmation dialogs. These are the places where language has the least space and the most responsibility.
Why Turkish Changes the Safety Risk
Turkish requires explicit relationships between action, object, condition, and consequence. English UI can often use compact phrases such as “confirm,” “clear,” “review,” “resume,” or “apply.” Turkish usually needs more context to prevent ambiguity.
A short Turkish verb may fit the UI but fail the workflow. For example, a button equivalent to “clear” may mean remove, reset, dismiss, clean, or erase depending on context. In medical software, those are not harmless differences.
Turkish also makes tone and responsibility visible. A warning that sounds too soft may not create enough caution. A warning that sounds too severe may interrupt workflow unnecessarily. A status message that sounds final may hide uncertainty.
This is why medical device UI localization must be reviewed with the clinical action in mind, not only the source string.
The Business Damage You Usually Misread
When Turkish medical software language is weak, the damage may appear as training difficulty, user hesitation, customer support questions, complaint patterns, or field feedback from distributors.
Teams may blame user education, interface complexity, or local workflow differences. Those may matter. But if the Turkish UI does not clearly guide action, training has to compensate for a localization problem.
That is inefficient and risky. Software should not need a trainer to explain what every Turkish warning means.
For manufacturers entering Türkiye, the Turkish interface also affects credibility. Hospitals, clinicians, distributors, and regulators notice whether the product feels controlled in the local language.
What Proper Turkish Medical Software Localization Does Instead
A proper workflow begins with context. Each string should be reviewed in its screen, user role, workflow step, and risk state. A button label in a routine setup screen does not behave like the same label in a clinical warning.
Terminology must align with the IFU, quick guides, training material, packaging, and support documentation. If a device function is named differently across the UI and IFU, users must reconcile the difference themselves. That should never happen.
Character limits should not force unsafe ambiguity. If a Turkish phrase needs more space to preserve meaning, the UI should be adjusted or supported with helper text. Safety should not be sacrificed to fit a button.
Independent linguistic review should check not only grammar but action clarity: what does the user think will happen after this interaction?
What to Audit Before Turkish Release
Start with all warnings, error states, confirmation dialogs, and action buttons. These are the highest-risk strings.
Then check measurement units, device modes, patient status labels, calibration steps, and result interpretation screens. Any term that affects clinical interpretation needs controlled translation.
Finally, review the complete ecosystem: software UI, IFU, training materials, support scripts, and release notes. The Turkish language layer should behave as one system.
This is where Turkish localization, medical translation, QA review, and terminology architecture must work together.
Where This Connects Inside the Turklingua Site
This topic supports the broader Turklingua authority cluster around Turkish localization, market-entry communication, quality assurance, and high-stakes content workflows.
- Turkish localization services
- Turkish marketing translation services
- Turkish translation quality assurance
- confidential project handling
Medical software does not fail only through code.
It can fail through one unclear instruction at the wrong moment.
A strong Turkish localization workflow protects the user, the manufacturer, and the clinical workflow.
The goal is simple: no hesitation where safety needs action.

Process authority: review terminology, tone, and decision logic before the market exposes the weakness.
FAQ
Why is medical software localization high risk?
Because software labels, alerts, buttons, and instructions can affect clinical behavior. A vague Turkish phrase may change how a user interprets a warning, procedure, or next action.
Is this different from IFU translation?
Yes. IFU translation is document-based. Software localization adds UI constraints, button labels, alerts, workflows, truncation risk, and real-time user decisions.
What should be checked before release?
Alerts, confirmation dialogs, action buttons, measurement labels, user roles, warnings, status messages, and linked IFU terminology should be reviewed as one controlled system.
Review the Turkish UI Before Safety Depends on It
We review Turkish medical software interfaces for action clarity, terminology control, clinical safety language, and user-facing risk.
Request Medical Software Localization Review